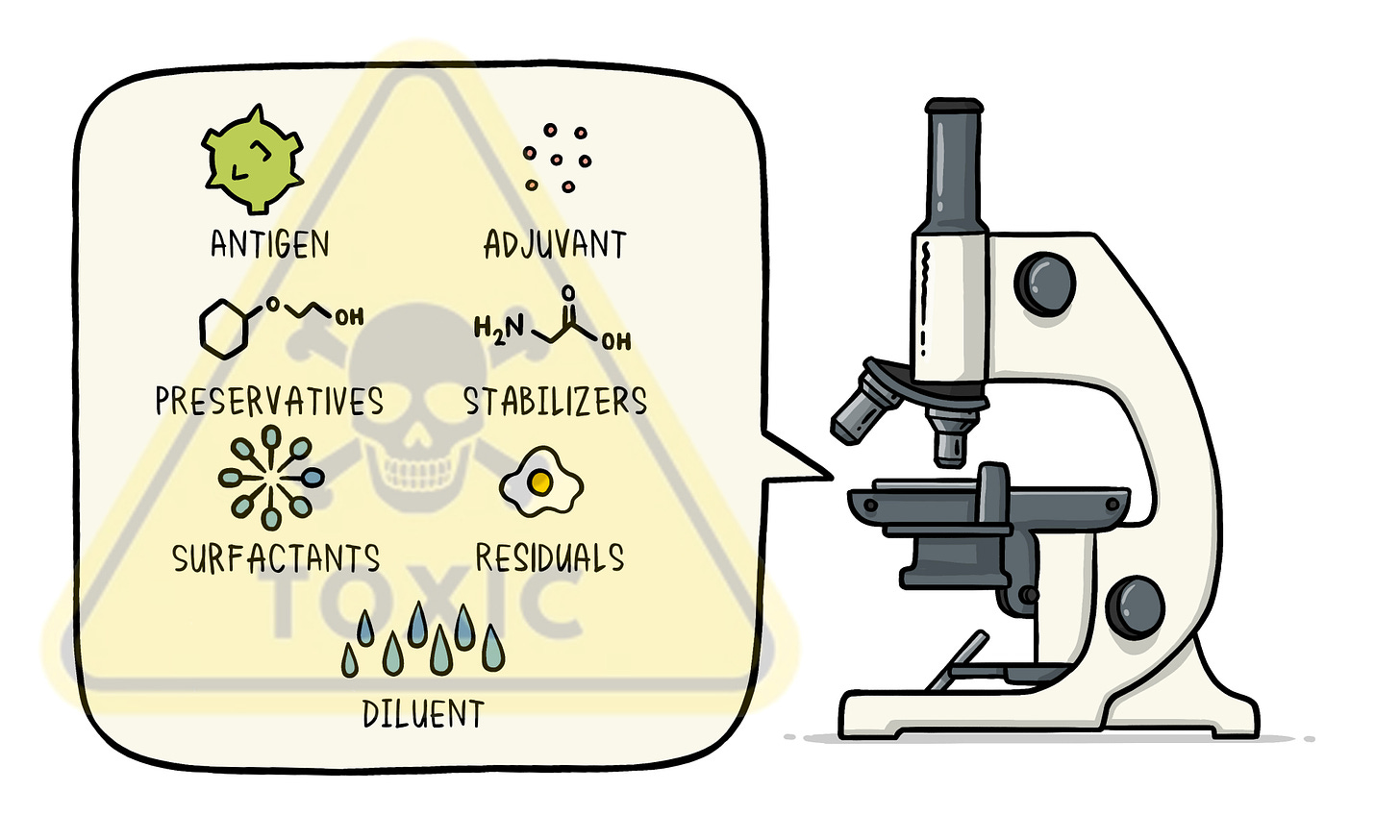
Many individuals overlook the composition of vaccines, presuming them to contain mere water or saline with virus fragments to provoke an immune response, aiding in disease prevention. However, the reality is more complex.
Understanding the contents of vaccines, particularly concerning injections into one's body or that of their children, should remain paramount in making informed decisions. Presented below are commonly used ingredients in vaccine production, alongside brief descriptions of associated risks.
Common Vaccine Ingredients
Inactivated or attenuated forms of viruses or bacteria: While these are used to stimulate an immune response, there's a slight risk of the vaccine causing the actual disease in rare cases, particularly in immunocompromised individuals.
Toxoids (inactivated toxins produced by bacteria): Although they're weakened or inactivated, there can still be risks of adverse reactions, including allergic reactions.
Aluminum salts (such as aluminum hydroxide, aluminum phosphate): Aluminum salts are used as adjuvants in vaccines to enhance the body's immune response. However, excessive aluminum exposure has been linked to potential neurological and developmental issues.
AS03 (squalene, DL-alpha-tocopherol, polysorbate 80): AS03 is an adjuvant used in some vaccines. Polysorbate 80 is known to cause allergic reactions and can also cross the blood/brain barrier, and squalene has been associated with autoimmune diseases in rare cases.
Thimerosal (ethylmercury): Thimerosal is a preservative that contains ethylmercury. While it's been largely removed from vaccines, its presence has raised concerns about mercury toxicity.
2-phenoxyethanol: This is a preservative used in some vaccines that may cause skin irritation and allergic reactions in sensitive individuals.
Formaldehyde: Formaldehyde is used to inactivate toxins in vaccines and prevent contamination. It's a known carcinogen.
Sugars (such as sucrose, lactose): Sugars are often used as stabilizers in vaccines. While they're generally safe, they may pose a risk to individuals with certain metabolic disorders.
Gelatin: Gelatin is used as a stabilizer in some vaccines and can cause allergic reactions, particularly in individuals with gelatin allergies.
Human serum albumin: This protein, derived from human blood, is used in some vaccines as a stabilizer and can pose a risk of allergic reactions or transmission of blood-borne diseases.
Sorbitol: Sorbitol is used as a stabilizer in some vaccines and can cause gastrointestinal upset in sensitive individuals.
Monosodium glutamate (MSG): MSG is used as a stabilizer in some vaccines and can cause allergic reactions in sensitive individuals.
Egg protein from chicken embryos: Some vaccines are produced using chicken eggs, which can pose a risk to individuals with egg allergies.
Antibiotics such as neomycin, streptomycin, polymyxin B: These antibiotics may be used during vaccine production and can cause allergic reactions in sensitive individuals.
Cell culture materials (from the medium used to grow viruses or bacteria during vaccine production): While rare, there's a potential risk of contamination from cell culture materials used during vaccine production.
This includes the use of aborted fetal tissue cells:
The cell lines derived from aborted fetal tissue that are used in vaccine production are WI-38 MRC-5, HEK-293, and PER.C6.
WI-38: This cell line was derived from the lung tissue of a female fetus aborted in 1962. It is commonly used in the production of vaccines such as rubella, varicella (chickenpox), and hepatitis A vaccines.
MRC-5: This cell line was derived from the lung tissue of a 14-week-old male fetus aborted in 1966. It is used in the production of vaccines against diseases like rubella, hepatitis A, varicella, and rabies.
HEK-293: The HEK-293 cell line was derived from human embryonic kidney cells obtained from a fetus aborted in the early 1970s. It is widely used in biotechnology and research, including the production of certain vaccines and therapeutic proteins.
PER.C6: The PER.C6 cell line was derived from human embryonic retinal cells obtained from an aborted fetus. It is primarily used in the production of viral vectors for gene therapy and vaccine development.
Traces of residual DNA and cellular proteins from these cell lines may be present in the final vaccine product.
Safe and Effective or Unavoidably Unsafe
While public health officials and doctors may try to reassure us that these ingredients are used in minuscule amounts and are harmless, scrutiny reveals a different reality.
Examining the questionable safety studies and extensive lists of potential adverse reactions listed in vaccine inserts, which often align with the known side effects of these ingredients, challenges the credibility of the "Safe and Effective" narrative.
Understanding the body's natural healing mechanisms underscores the fact that we are not inherently deficient in any of these potentially toxic substances. Injecting them into the body raises significant ethical and health concerns. It's a scenario where pharmaceutical companies seem to create the problem to offer the solution. Vaccines, in essence, create lifelong customers for the medical industry.
When it comes to vaccines and being injured there is virtually no recourse. The liability falls solely on the individual. Think long and hard about the decision to inject these ingredients into the body. You’ll never regret doing the research and making an informed decision.
Click the download button below for a PDF of the CDC Vaccine Excipient Summary.